Process Visualization with Remote Monitors in Pharmaceutical Cleanrooms

Application
Vaccine production in the pharmaceutical industry requires strict hygiene standards for both personnel and equipment. To ensure quality in this sector, several organizations like the European Medicines Agency (EMA), the American Food and Drug Administration (FDA) and the WHO release Good Manufacturing Practice (GMP) guidelines for their scope of countries that all companies must follow. The following API (Active Pharmaceutical Ingredient) application is from a biopharmaceutical company with grade B and C cleanroom spaces. The company uses two application programs in production: one manufacturing execution system (MES) and one distributed control system (DCS).
Goal
The company needs operator workstations for monitoring and controlling production processes installed directly in its production area. To comply with GMP standards, the workstations must be resistant to chemicals and cleaning agents and the surface finish must be easy to clean. Due to a limited amount of available space at the plant, each of the 30 workstations in the production area should be able to display both applications (DCS and MES) on one remote monitor.
Solution

The stainless steel housing is designed for easy cleaning.
Specially designed for GMP applications, VisuNet GMP operator workstations from Pepperl+Fuchs are perfectly tailored to the requirements of visualization systems in the pharmaceutical industry. The surface of the slim stainless steel housing is designed for easy cleaning, with no gaps or areas where liquid can accumulate. The stainless steel enclosure has a finish with a surface roughness of 0.8 μm. All materials are tested for their resistance to a wide range of chemicals and cleaning agents. To accommodate the dual application on one VisuNet Remote Monitor, a new firmware function, MSS (Multiple Simultaneous Sources), was developed. This feature ensures both applications from different servers are quickly accessible and easily displayed full-screen on the monitor by pressing a key combination.
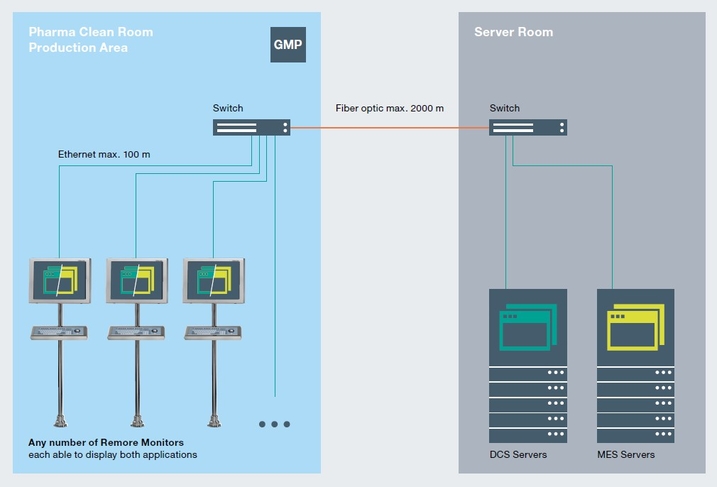
DCS and MES can both be accessed quickly from one remote monitor.
Benefits
With Pepperl+Fuchs, the pharmaceutical company receives a complete, GMP-compliant solution from a single source. The custom-designed MSS software made it is possible for the customer to operate both the DCS and MES applications from one VisuNet Remote Monitor, saving precious space in the production area. Other custom modifications, such as an additional mount for connecting a radio barcode scanner or a country-specific keyboard layout, are easy due to the VisuNet GMP’s modular concept. In addition to control in the production area, remote monitors can also be managed remotely via network from outside the cleanroom with VisuNet Control Center software.
At a glance
- Production monitoring and control with the VisuNet GMP in grade B and C spaces
- Modular concept of VisuNet GMP allows customer-specific design
- Remote management via network using VisuNet Control Center software
- Complete solution from a single source







 +65 6779 9091
+65 6779 9091